 
For sodium chloride, Δ H lattice = 769 kJ. In both cases, a larger magnitude for lattice energy indicates a more stable ionic compound. Thus, make sure to confirm which definition is used when looking up lattice energies in another reference. Another way is to use an equivalent but opposite convention, wherein the lattice energy is exothermic (negative values) and described as the energy released when ions combine to form a lattice. Here, the convention is used where the ionic solid is separated into ions, meaning the lattice energies will be endothermic (positive values). For the ionic solid sodium chloride, the lattice energy is the enthalpy change of the process: The lattice energy (Δ H lattice) of an ionic compound is defined as the energy required to separate one mole of the solid into its component gaseous ions. The lattice energy of a compound is a measure of the strength of this attraction. Thus, the magnitude of lattice energy is directly proportional to the product of the ion charges and is inversely proportional to the distance between the ions.Īn ionic compound is stable because of the electrostatic attraction between its positive and negative ions. This is why it takes nearly four times as much energy to separate the calcium oxide into gaseous ions as compared to sodium fluoride. However, calcium and oxide ions are divalent and the product of their charge is 4, which is four times greater than sodium fluoride. 
The ionic distance in both compounds is similar, but the lattice energy of calcium oxide is almost four times greater than sodium fluoride.īoth sodium and fluoride are monovalent ions the product of their charge is 1. According to Coulomb’s law, the potential energy of ions is directly proportional to the product of their charges.Ĭonsider two ionic compounds sodium fluoride and calcium oxide. In addition to the ionic radius, the magnitude of lattice energy also depends on the ion charges. Thus the lattice energy to separate solid lithium bromide is larger than for potassium bromide being +807 kJ/mol versus +682 kJ/mol, respectively. For example, the bond length of lithium bromide and potassium bromide differs between 217 pm and 282 pm, respectively.ĭue to the increased internuclear distance, the attraction between the ions decreases and it becomes much easier to separate the ions. As the size of the metal ion increases, the distance between ions, or the bond length, becomes larger as well. In the periodic table, the ionic radius of the alkali and alkaline earth metals increases down the column. 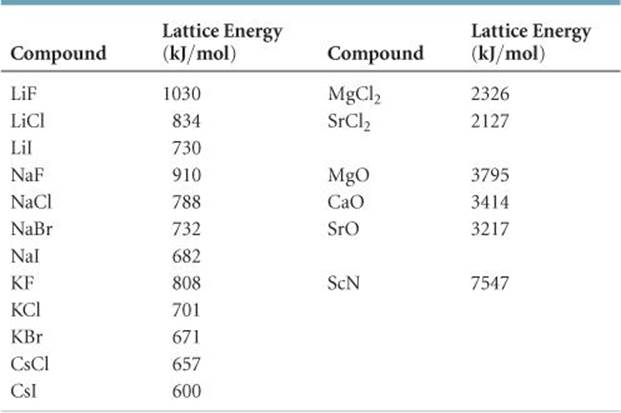
Why does each ionic compound have a different lattice energy, and which factors does it depend on?Īn ionic compound consists of an orderly arrangement of a large number of charged ions attracted to each other by electrostatic interactions.Īccording to Coulomb’s law, the potential energy of two ions is inversely proportional to the distance between the ions, which in turn depends on the ionic radius. Yet, when sodium chloride or magnesium oxide form, their lattice energy differs significantly. Therefore, CaCl₂ would have the largest lattice energy.Lattice energy is associated with the formation or separation of an ionic lattice. So, CsBr₂ will have less lattice energy.ĬaCl₂ has + 2 charge on calcium as well as the ionic radius is also samll. CsBr₂ has +2 charge on calcium but the size of both ios is larger. So they have less lattice energy.ĬsI ionic solid have a larger size of ions Cs⁺ and I⁻. Lattice energy is directly proportional to the charge on ions and inversely proportional to the interionic distance between ions.įor NaCl and NaF the charge on the ions is +1 and -1 while the size of the ions is small. The relation between lattice energy and lattice enthalpy at constant pressure is given by: Lattice energy is the heat of formation when one mole of crystalline ionic solid is generated from its constituent ions in their gaseous state.įor example, the lattice energy of sodium chloride crystal is the energy change of reaction when Na⁺ and Cl⁻ ions react to produce a NaCl crystal, which is equal to -786 kJ/mol. The ionic solid CaCl₂ would have the largest lattice energy.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
